 
However, the patient’s particular medical condition may dictate whether or not a subcutaneous, chronically implanted device can be tolerated. The Patient Assistant activates the data management feature in the Reveal™ insertable cardiac monitor to initiate recording of cardiac event data in the implanted device memory.Ĭontraindications: There are no known contraindications for the implant of the Reveal LINQ ICM or for the Reveal LINQ Mobile Manager system. Patient Assistant: The Patient Assistant is intended for unsupervised patient use away from a hospital or clinic. 
The patient connector is intended to be used by healthcare personnel only in a clinical or hospital environment. Cardiac monitor that contiusly monity st elevation Bluetooth#The patient connector uses Bluetooth ® technology to transmit implantable heart device data to the Reveal LINQ Mobile Manager app for further processing. Cardiac monitor that contiusly monity st elevation portable#The Medtronic patient connector is a portable electronic device using low frequency inductive telemetry to communicate with the Reveal LINQ ICM. Reveal LINQ Mobile Manager System: The Reveal LINQ Mobile Manager app is intended for programming and interrogating the Reveal LINQ ICM LNQ11. The device has not been tested specifically for pediatric use. 
Patients who experience transient symptoms such as dizziness, palpitation, syncope, and chest pain, that may suggest a cardiac arrhythmia.Patients with clinical syndromes or situations at increased risk of cardiac arrhythmias.Indications: The Reveal LINQ™ insertable cardiac monitor (ICM) is an implantable patient-activated and automatically activated monitoring system that records subcutaneous ECG and is indicated in the following cases: Reveal LINQ Insertable Cardiac Monitor, Reveal LINQ Mobile Manager System, and Patient Assistant For further information, please call Medtronic at (800) 328-2518 (Technical Services), (800) 551-5544 (Patient Services), and/or consult Medtronic’s website at Caution: Federal law (USA) restricts prescription devices to sale by or on the order of a physician. Cardiac monitor that contiusly monity st elevation manuals#See the device manuals for detailed information regarding the implant procedure, indications / intended use, contraindications, warnings, precautions, and potential complications/adverse events. There are no known adverse events associated with the use of any LINQ II ICM wireless accessory. Potential adverse events from the LINQ II ICM include, but are not limited to, device rejection phenomena (including local tissue reaction), device migration, infection, and erosion through the skin. See product manuals for details and troubleshooting instructions. Wireless accessories available for use with LINQ II may experience connectivity or performance issues. MRI scans should be performed only in a specified MR environment under specified conditions as described in the LINQ II MRI Technical Manual. Patients with the LINQ II ICM should avoid sources of diathermy, high sources of radiation, electrosurgical cautery, external defibrillation, lithotripsy, therapeutic ultrasound and radiofrequency ablation to avoid electrical reset of the device, and/or inappropriate sensing as described in the Medical procedure and EMI Warnings, Precautions and Guidance Manual. However, the patient’s particular medical condition may dictate whether or not a subcutaneous, chronically inserted device can be tolerated. 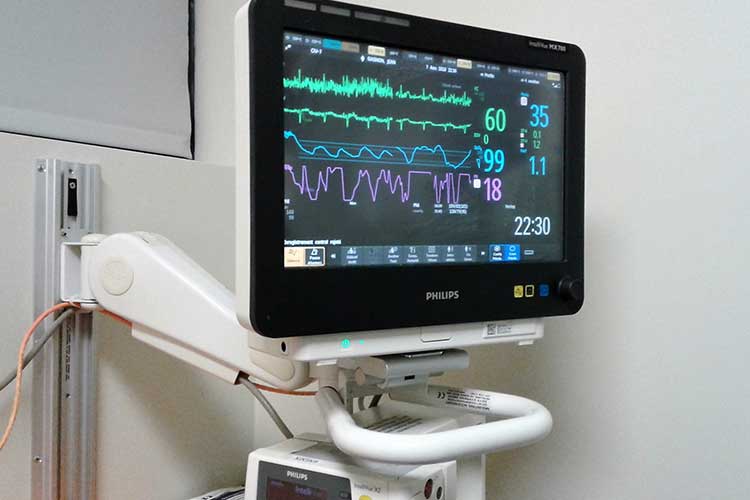
There are no known contraindications for the insertion of the LINQ II ICM or its accessories.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
